Abstract
Cite
- MLA: Sultana, S.; Alimullah, M.; Tazim, M. A.; Ghosh, H. C.; Fauzia, F.; Akhter, T.; Zaman, O.; Hossen, M. T.; Subhan, N., Alam, M. A.. "A Review on Phytochemistry and Pharmacological Potential of Sonneratia apetala (Keora): A Mangrove Plant of the Sundarbans." J. Bio. Exp. Pharm 3.2 (2025): 1-23.
- APA: Sultana, S.; Alimullah, M.; Tazim, M. A.; Ghosh, H. C.; Fauzia, F.; Akhter, T.; Zaman, O.; Hossen, M. T.; Subhan, N., Alam, M. A., (2025). A Review on Phytochemistry and Pharmacological Potential of Sonneratia apetala (Keora): A Mangrove Plant of the Sundarbans. J. Bio. Exp. Pharm, 3(2), 1-23.
- Chicago: Sultana, S.; Alimullah, M.; Tazim, M. A.; Ghosh, H. C.; Fauzia, F.; Akhter, T.; Zaman, O.; Hossen, M. T.; Subhan, N., Alam, M. A.. "A Review on Phytochemistry and Pharmacological Potential of Sonneratia apetala (Keora): A Mangrove Plant of the Sundarbans." J. Bio. Exp. Pharm 3, no. 2 (2025): 1-23.
- Harvard: Sultana, S.; Alimullah, M.; Tazim, M. A.; Ghosh, H. C.; Fauzia, F.; Akhter, T.; Zaman, O.; Hossen, M. T.; Subhan, N., Alam, M. A., 2025. A Review on Phytochemistry and Pharmacological Potential of Sonneratia apetala (Keora): A Mangrove Plant of the Sundarbans. J. Bio. Exp. Pharm, 3(2), pp.1-23.
- Vancouver: Sultana, S.; Alimullah, M.; Tazim, M. A.; Ghosh, H. C.; Fauzia, F.; Akhter, T.; Zaman, O.; Hossen, M. T.; Subhan, N., Alam, M. A.. A Review on Phytochemistry and Pharmacological Potential of Sonneratia apetala (Keora): A Mangrove Plant of the Sundarbans. J. Bio. Exp. Pharm. 2025;3(2):1-23.
Keywords
1. Introduction
2. Methodology
To locate the details on phytochemistry and pharmacological activities of Sonneratia apetala, the literature works were comprehensively searched for relevant studies. The search strategy involved searching keywords, namely, Sonneratia apetala”, “Keora”, “mangrove medicinal plant,” “antioxidant activity,” “antidiabetic,” “hepatoprotective,” and “reno-protective,” from PubMed, Scopus, Web of Science, ScienceDirect, and Google Scholar.
After reviewing in vitro, in vivo, and pharmacological research articles for the past decade, full-text articles containing information on S. apetala's phytochemical ingredients, in vitro tests, in vivo models, or mechanism perspectives were obtained only if relevant. The criteria for exclusion included papers with inadequate mechanisms, unrelated mangrove species, or solely ecological studies lacking sufficient pharmacological data. Hence, this review seeks to collect phytochemical and pharmacological information on Sonneratia apetala, presenting an overview of its therapeutic potential and insights for future clinical research.
3. Sonneratia apetala: Mangrove Apple
Sonneratia apetala, with a family of Lythraceae is a mangrove plant that is expanding rapidly and widely found in the tropical coastal regions, especially in the Sundarbans Forest of Bangladesh, India, Malaysia, Australia, China, Myanmar, New Guinea, and other countries [6, 8-10]. It is known by several names in different countries such as Mangrove Apple, Motitavar, Keora, Kandal, Chipi, and Keruan [10]. The fruits of this species are frequently used as food, nutrition, and medicine to treat a variety of ailments [11]. The plant showed antibacterial, cytotoxic, antiviral, antioxidant, and antidiabetic activity[8, 12]. There are many active compounds in the fruit of the plant.
4. Taxonomic Classification of S. apetala
Kingdom: Plantae; Subkingdom: Viridiplantae; Infrakingdom: Streptophyta; Phylum (Division): Tracheophyta; Subphylum (subdivision): Spermatophytina; Class: Magnoliopsida; Superorder: Rosanae; Order: Myrtales; Family: Lythraceae; Genus: Sonneretia; Species: Sonneretia apetala [10] [13]
5. Phytochemistry of S. apetala
The ability of Keora to survive and thrive in harsh, high salinity environment of mangrove forests is directly attributed to its unique and abundant profile of defensive secondary metabolites. These compounds are synthesized by the plants to combat and counteract the severe osmotic and oxidative stress given to it by its habitat [4]. Chemical screening across various plant parts consistently revealed the presence of major classes of bioactive compounds, primarily polyphenols and flavonoids, alongside significant quantities of triterpenoids, alkaloids, and tannins. The high concentration of these metabolites is the pharmacological basis for all the biological activities discussed in this review.
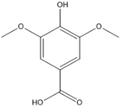
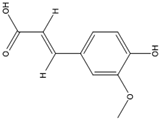
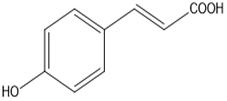
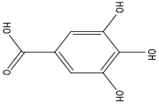
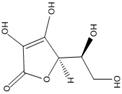
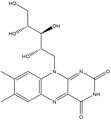
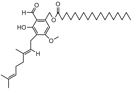
Key compounds identified through isolation and analysis include the potent polyphenols gallic acid and ellagic acid, the triterpenoids lupeol and betulinic acid, and the flavonoids quercetin and catechin. Notably, seeds and fruit pericarp are typically reported as being especially rich in polyphenols and flavonoids. A detailed, compound-by-compound breakdown of the phytochemicals isolated from S. apetala, along with their specific plant source and corresponding literature references, is presented in Tables 1 and 2.
Table 1: Chemical Constituents of the fruit of S. apetala
|
Compounds |
Activity |
Structure |
Ref. |
|
|
Phenolic compounds
|
Caffeic acid |
Antioxidant, anti-inflammatory, anticarcinogenic. |
|
[14, 15] |
|
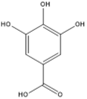
Syringic acid |
Antioxidant, anti-inflammatory, antiproliferative, anticancer, antimicrobial, antiendotoxic. |
|
[14, 16] |
|
|
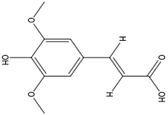
Ferulic acid |
Antioxidants, anti-inflammatory, anticarcinogenic, hepatoprotective, and antibacterial. |
|
[14, 17] |
|
|
p-Coumaric acid |
Antioxidant, anti-bacterial, antitumor, anti-inflammatory. |
|
[14, 18] |
|
|
Gallic acid |
Antioxidant, anti-inflammatory, antineoplastic. |
|
[14, 19] |
|
|
Sinapic acid |
Antioxidant, antimicrobial, anti-inflammatory, anticancer. |
|
[14, 20] |
|
|
Coumarin |
Anticoagulant, antioxidant, anti-inflammatory, antitumor, antiviral, antibacterial. |
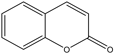 |
[14, 21] |
|
|
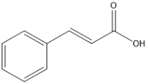
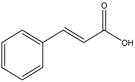
trans-Cinnamic acid |
Antioxidant, antibacterial, anti-inflammatory, antitumor. |
|
[7, 22] |
|
|
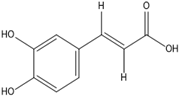
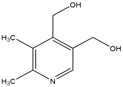
Phenol, 3,5-bis(1,1-dimethylethyl) |
Anticancer, antioxidant, antimicrobial. |
|
[7, 23] |
|
|
|
Benzenepropanoic acid |
Antioxidant, anti-inflammatory. |
|
[14] |
|
Flavonoids |
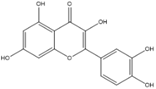
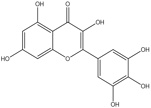
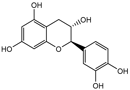
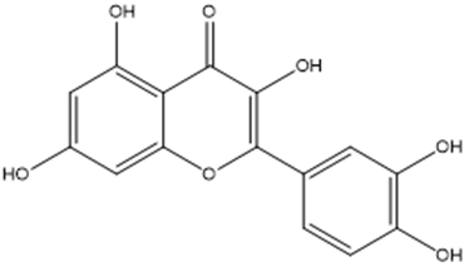
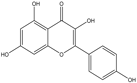
Quercetin |
Anticancer, antioxidant, anti-inflammatory, anti-cardiovascular, anti-aging, neuroprotective. |
|
[14, 24] |
|
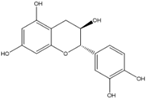
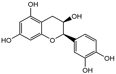
Catechin |
Anticancer, antioxidant, anti-inflammatory. |
|
[14, 25] |
|
|
Rutin |
Antioxidant, anti-inflammatory, anti-proliferative. |
|
[14, 26] |
|
|
Myricetin |
Antioxidant, anticancer, antidiabetic and anti-inflammatory. |
|
[14, 27] |
|
|
Apigenin |
Antiproliferative, anti-inflammatory, and antimetastatic |
|
[14, 28] |
|
|
Kaempferol |
Antioxidant, anti-inflammatory, anticancer, antidiabetic, cardioprotective, neuroprotective. |
|
[7, 29] |
|
|
Triterpenoids |
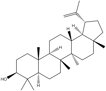
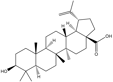
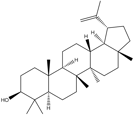
Lupeol |
Anti-inflammatory, antiprotozoal, hepatoprotective, cancer preventive.
|
|
[14, 30] |
|
|
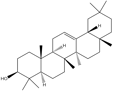
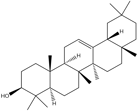
β-amyrin |
Antioxidant. |
|
[14, 31] |
|
Others |
1,2-benzene dicarboxylic acid ester |
Anticancer, antibacterial, antidiabetic. |
|
[7] |
|
2-methyltetracosane |
Antibacterial, antioxidant. |
|
[7, 32] |
|
|
Tetratetracontane |
Antioxidant, cytoprotective, hypoglycemic, hypolipidemic, antibacterial. |
|
[7, 33] |
|
|
Heptacosane |
Antibacterial, antifungal. |
|
[7] |
|
|
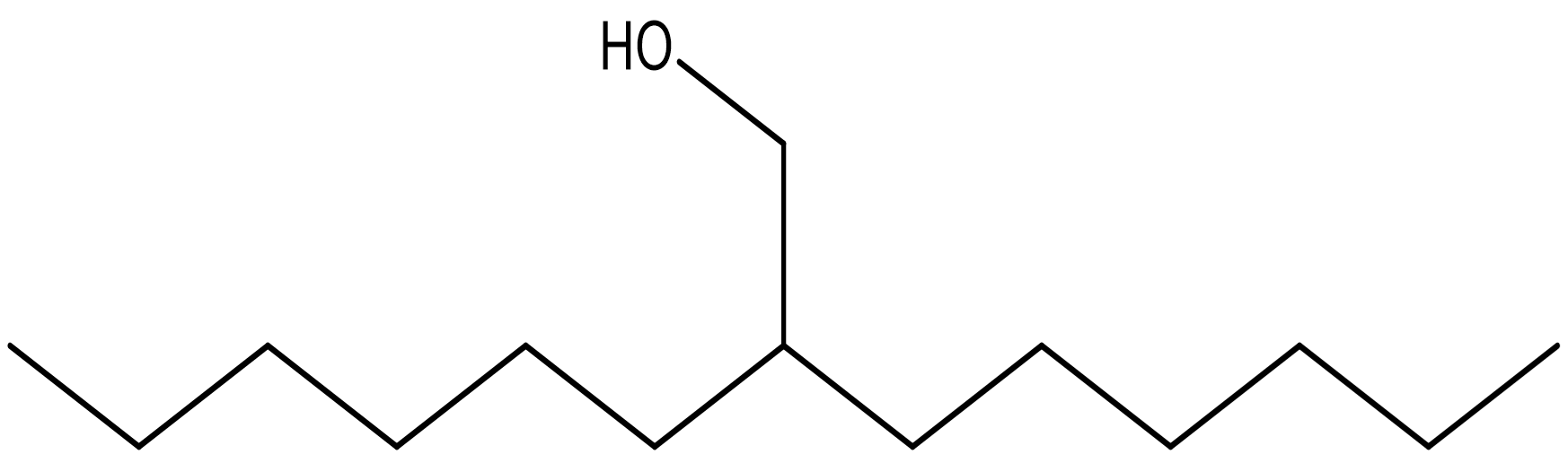
2-hexyl-1-octanol |
Antimicrobial. |
|
[7] |
|
|
Vitamin |
Ascorbic acid |
Antioxidant, anti-inflammatory. |
|
[14] |
|
|
Vitamin B2 |
Antioxidant, |
|
[14] |
|
Vitamin B5 |
Antioxidant, anti-inflammatory. |
|
[14, 34] |
|
|
Vitamin B6 |
Anti-inflammatory. |
|
[14, 35] |
Table 2: Chemical constituents of the seed of S. apetala
|
Compounds |
Activity |
Structure |
Ref. |
|
|
Polyphenols
|
Caffeic acid |
Antioxidant, anti-inflammatory, anticarcinogenic. |
|
[6, 15] |
|
(+)-catechin |
Anticancer, antioxidant, anti-inflammatory, and anti-allergy. |
|
[6, 25] |
|
|
(-)-epicatechin |
Antioxidant, anti-inflammatory, antimicrobial, antitumor, cardioprotective. |
|
[6, 36] |
|
|
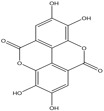
Ellagic acid |
Antioxidant, and antiadipogenic, anticancer. |
|
[6, 37] |
|
|
Gallic acid |
Antioxidant, anti-inflammatory, antineoplastic. |
|
[6, 19] |
|
|
Quercetin |
Anticancer, antioxidant, anti-inflammatory, cardioprotective, anti-aging, and neuroprotective. |
|
[6, 19] |
|
|
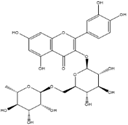
Rutin hydrate |
Antioxidant, anti-inflammatory, anti-proliferative. |
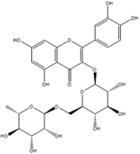 |
[26, 38] |
|
|
trans-ferulic acid |
Antioxidant |
|
[38] |
|
|
trans-cinnamic acid |
Antioxidant, antibacterial, anti-inflammatory, antitumor. |
|
[22, 38] |
|
|
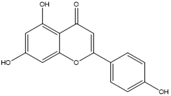
Myricetin |
Antioxidant, anticancer, antidiabetic, and anti-inflammatory. |
|
[38] |
|
|
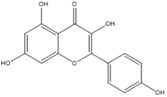
Kaempferol |
Antioxidant, anti-inflammatory, anticancer, antidiabetic, cardioprotective, neuroprotective. |
|
[30, 38] |
|
|
Fatty Acids |
Margaric acid |
Antitumor, antimicrobial. |
|
[39, 40] |
|
8,11-otadecadienoic acid, methyl ester |
Antimicrobial . |
|
[40, 41] |
|
|
Steric acid, methyl ester |
Antibacterial and antifungal. |
|
[40, 42] |
|
|
Linoleic acid, methyl ester |
Antioxidant. |
|
[43] |
|
|
Oleic acid, methyl ester |
Acaricidal, antimicrobial. |
|
[43, 44] |
|
|
Oleic acid |
Antifungal, antitumor. |
|
[45] |
|
|
Arachidic acid |
Anti-inflammatory, cardioprotective, anticlotting. |
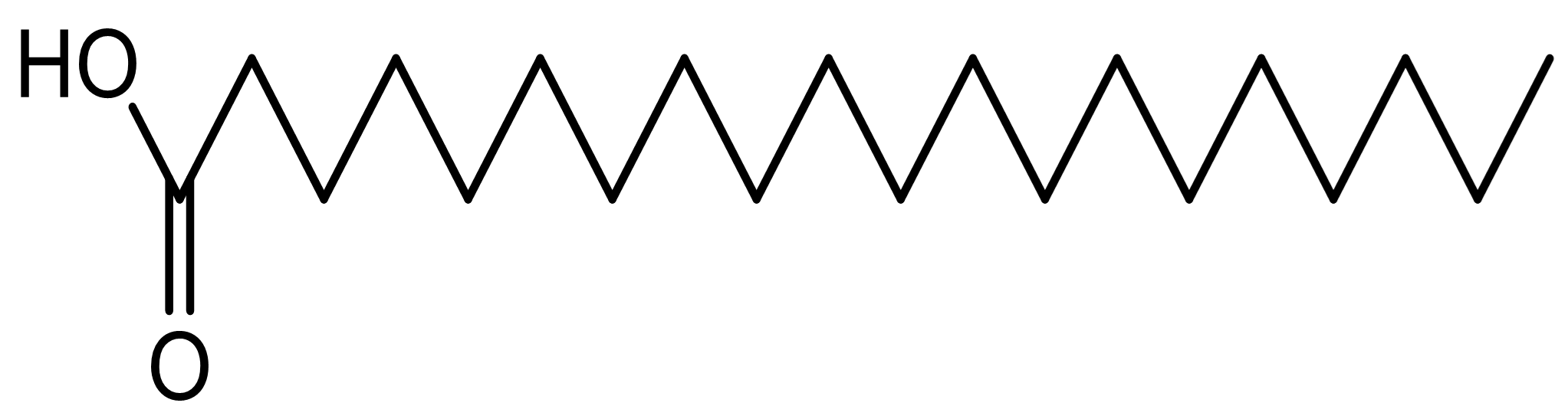 |
[40, 60] |
|
|
Linoleic acid |
Anti-inflammatory, anticoagulant, cardioprotective. |
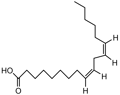 |
[40, 47] |
|
|
Stearic acid |
Neuroprotective, antioxidant, antilipidemic. |
[40, 48] |
||
|
Tetracosanoic acid |
Antioxidant, anticancer, antihypertensive, anti-inflammatory |
[40, 49] |
||
| Palmitic acid | Antioxidant, anticancer, antihypertensive, anti-infalmmatory | [12] | ||
|
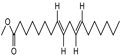
Ester |
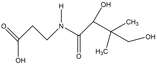
bis(2-ethylhexyl) ester |
Antimutagenic. |
|
[38, 50] |
|
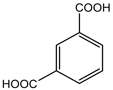
1,3-benzenedicarboxylic acid |
Catalyst. |
|
[38] |
|
|
Triterpenoid |
Lupeol |
Anti-inflammatory, anticancer, antimicrobial. |
|
[10] |
|
β-amyrin |
Antioxidant. |
|
[10, 31] |
|
|
Lupeone |
Anti-inflammatory, antiprotozoal, hepatoprotective, |
|
[10] |
|
|
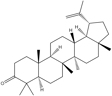
Betulinic acid |
Anti-inflammatory, antibacterial, antiviral, antidiabetic, antimalarial, antitumor |
|
[10, 52] |
|
|
Steroid |
Stigmast-5-ene 3beta |
Antidiabetic. |
|
[10, 51] |
|
Others |
Physcion |
Anti-proliferative. |
|
[10, 53] |
|
Gibberellin |
Anti-inflammatory. |
|
[10, 54] |
6. Pharmacological activities of S. apetala
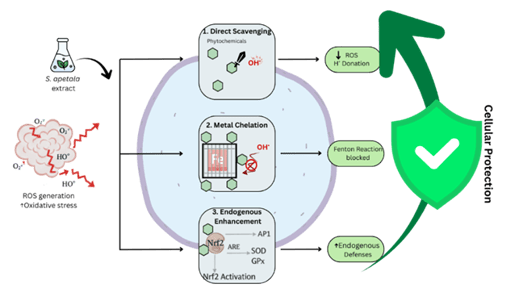
Figure 2: Generalized mechanism of plant-derived antioxidant action
The imbalance between the production of reactive oxygen species (ROS) and the body’s antioxidant defenses is termed as Oxidative stress and it is a major factor in the development of chronic disorders like diabetes, liver damage, renal dysfunction [55]. Due to the multi-targeting potential of plant-based medicines, natural antioxidants have drawn a lot of interest as possible treatment for these disorders [56]. Several studies have already evaluated the antioxidant activity of Sonneratia apetala based on both in vitro and in vivo models.
DPPH (2,2-diphenyl-1-picrylhydrazyl), ABTS (2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)), FRAP (Ferric reducing antioxidant power) and other in vitro studies have revealed a strong dose-dependent antioxidant activity in the extracts of Sonneratia apetala, thus demonstrating its free radical scavenging potential [57]. In vivo studies provided further evidence of the antioxidant activities of Sonneratia apetala extracts as administering its extracts to experimental animal models showed an increase in the activities of antioxidant enzymes like glutathione peroxidase (GPx), catalase (CAT) and Superoxide dismutase (SOD). At the same time, it lowered lipid peroxidation indicators like malondialdehyde (MDA), suggesting that the tissues were protected from oxidative damage [58].
Polyphenols and other bioactive compounds are mainly responsible for these antioxidant effects as they can usually work in three ways: they scavenge the free radicals by giving them hydrogen atoms or electrons, chelate Fe2+ ions to stop the Fenton reaction from forming hydroxyl radicals, modulation of the expression of antioxidant enzymes to reinforce cellular defense [59]. The antioxidant activities of Sonneratia apetala observed throughout various studies are summarized in Table 3, which shows the types of extracts tested, experimental models used, assays employed and key outcomes of the tests. Overall, the compiled evidences demonstrated that S. apetala showed significant antioxidant potential across multiple experimental systems, providing a strong support for its antioxidant properties.
Table 3: Antioxidant properties of Sonneratia apetala extracts
|
Model |
Treatment |
Result |
Ref. |
|
In vitro. DPPH, NO free radical scavenging assay. |
Pericarp methanolic extract. |
Showed free radical scavenging activity. |
[8] |
|
In-vitro. Reducing power and DPPH radical scavenging activity. |
Aqueous extract of fruit. |
Showed high reducing power and DPPH radical scavenging activity. |
[60] |
|
In-vitro. Colorimetric method- Fe-chelating activity. |
Fruit extract of n-hexane, chloroform, and methanol. |
All fractions showed chelating activity. Methanolic fraction showed the highest activity with IC50 of 165 μg/mL. |
[61] |
|
In-vivo. Male Swiss albino mice- 100mg/kg I.P. injection of ferric carboxymaltose induced oxidative stress. |
Methanol and n-hexane extract of fruit at 250, 500, 750, and 1000 μg/kg for 21 days. |
Reduced iron profile; highest methanolic dose completely ameliorated blood and liver iron overload and prevented oxidative stress. |
[61] |
|
In-vitro. DPPH, ABTS, and NO radical scavenging activity. |
Ethanolic fruit extract. |
Showed strong scavenging activity against ABTS, DPPH and NO radicals. |
[14] |
|
In vitro. DPPH, NO, and ABTS scavenging; In vivo. Oxidative stress mice model. |
Various fruit extracts (Pericarp methanolic, Aqueous, n-hexane, chloroform, methanol, and ethanolic). |
Extracts showed radical scavenging, reducing power, and chelating; CHCl₃ excellent (IC₅₀ 13.76 μg/mL), n-Hex & EtOAc moderate (IC₅₀ 42.03 & 49.998); highest methanol dose prevented oxidative stress in iron-overloaded mice |
[62] |
|
In vitro. DPPH free radical scavenging method. |
Methanol extract of leaf at concentrations of 5−80 μg/mL. |
strong antioxidant activity (IC50 of 41.92 μg/mL). |
[63] |
|
In vitro assays including DPPH scavenging ability, NO free radical scavenging ability, and metal chelating ability |
Methanol extracts of the fruit's seeds and pericarp |
Seed extract showed stronger antioxidant activity than pericarp, with methanolic extract being the most effective. |
[38] |
|
In vitro: DPPH, NO free radical scavenging assay. |
Pericarp methanolic extract. |
Showed free radical scavenging activity |
[64] |
|
In-vitro: Reducing power and DPPH radical scavenging activity |
Aqueous extract of fruit. |
Showed high reducing power and DPPH radical scavenging activity |
[64] |
|
In vitro: Colorimetric method - Fe-chelating activity. |
Fruit extract of n-hexane, chloroform, and methanol. |
All fractions showed chelating activity. Methanolic fraction showed the highest activity with Ic50 of 165 μg/mL |
[64] |
|
In-vivo: Male Swiss albino mice - 100mg/kg I.P. injection of ferric carboxymaltose induced oxidative stress. |
Methanol and n-hexane extract of fruit at 250, 500, 750, and 1000 μg/kg for 21 days. |
Reduced iron profile; highest methanolic dose completely ameliorated blood and liver iron overload and prevented oxidative stress |
[64] |
|
In vitro antioxidant activity using DPPH and FRAP assays |
Methanolic, ethanolic and aqueous extract of leaf |
Methanolic leaf extract showed 77.37% scavenging, ethanolic 75.14%, and aqueous 68.12% |
[65] |
|
In vitro DPPH, reducing power, and total antioxidant capacity assays |
Aqueous extract of S. apetala fruit powder |
Strong antioxidant activity: IC₅₀ = 33.5 µg/mL (DPPH), reducing power = 170.83 mg GAE/g, total capacity = 210.43 mg AAE/g. |
[66] |
|
In vivo. PO/HX-induced hyperuricemic mice. |
Aqueous extract of leaves, further concentrated with 60% ethanol |
Restored renal SOD, CAT, GSH-Px and reduced MDA and ROS in kidney tissue; ↓UA, BUN, CRE, Cys-C; |
[58] |
|
In vitro (DPPH, ABTS⁺, NO, superoxide, hydroxyl radical scavenging, Reducing power) |
Hydro-methanolic (20:80) extract of S. apetala leaves |
strong in vitro antioxidant activity, effectively scavenging |
[67] |
|
In vivo (adult male Wistar albino rats) |
Hydro-methanolic extract of Sonneratia apetala leaves |
Exhibited antioxidant effects in gastric tissue by reducing lipid peroxidation and enhancing levels of glutathione and catalase |
[67] |
|
In vitro (chemical assays: DPPH, ABTS⁺, NO, O₂⁻, HO•, Reducing power |
Aqueous extract of leaves and branches |
Showed antioxidant activity (e.g., DPPH IC₅₀ = 0.81 mg/mL, ABTS⁺ IC₅₀ = 0.16 mg/mL, others) |
[68] |
|
In vivo (Kunming mice, HUA model) |
Aqueous extract of leaves and branches (50, 100, 200 mg/kg) for 7 days |
Dose-dependent ↑ SOD, CAT, GSH; ↓ MDA, ROS in kidney; strongest effect at 200 mg/kg, surpassing BZM (CAT/GSH) and FBX (GSH); indicates potent antioxidant activity |
[68] |
|
In vitro. DPPH, total phenolic, total flavonoid, and total antioxidant capacity assays. |
Methanolic leaf extract of Sonneratia apetala |
Exhibited antioxidant activity |
[69] |
|
In vitro. DPPH, H₂O₂, hydroxyl, and superoxide radical scavenging assays; total phenolic, flavonoid, and tannin content. |
Ethanolic extract of pneumatophore (aerial root). |
Strong free radical scavenging activity (IC₅₀: DPPH 71.77 µg/ml, H₂O₂ 97.27 mg/L, OH 79.62 mg/L, O₂⁻ 108.89 mg/L |
[70] |
|
In vivo. Oral glucose tolerance test (mice). |
Ethanolic extract of pneumatophore at 250 & 500 mg/kg bw; |
Significantly reduced blood glucose at 60 and 120 min; effect comparable to standard drug |
[70] |
|
In vitro. DPPH, nitric oxide (NO) free radical scavenging assay |
Methanol fraction of seeds (MeS) |
Strong antioxidant activity from polyphenols and vitamin C. |
[12] |
|
In vitro. Lipid-soluble antioxidant assay |
n-Hexane fraction of seeds (HS) |
Antioxidant activity from lipophilic compounds (ascorbyl palmitate, fatty acids). |
[12] |
|
In vitro antioxidant assays (DPPH, free radical scavenging) |
Leaf extracts (Hexane, Ethyl acetate, Methanol) |
Methanol extract showed strongest activity; ethyl acetate moderate; hexane weak. |
[71] |
|
In vitro. Total phenolic content, DPPH free radical scavenging assay |
Crude extract, ethanol fraction, acetone fraction of S. apetala pneumatophores |
Acetone fraction showed strongest antioxidant activity (IC50 2.4 μg/mL), ethanol fraction very low, crude extract moderate.
|
[72] |
|
In vitro (DPPH, NO scavenging, reducing power, Fe²⁺ chelation, TAC) |
Methanol, diethyl ether, chloroform, and ethyl acetate fractions of Sonneratia apetala seeds |
Exhibited strong to moderate antioxidant activity; methanol seed fraction (MS) showed highest activity |
[6] |
|
In vitro: DPPH, reducing power, total antioxidant capacity (TAC), total phenolic content (TPC) |
Seeds and pericarps; fresh and stored; uncooked or cooked (2–20 min); methanol extract of seeds |
Antioxidant property: Seeds > pericarps; cooking seeds 20 min and pericarps 5–10 min gave highest activity; fresh > stored; methanol seed extract strongest |
[73] |
|
In vitro (DPPH, reducing power, Fe2+ chelation, TPC) |
Methanolic extract of bark |
methanolic bark extract showed strong antioxidant activity |
[57] |
|
In vitro antioxidant assays (DPPH, ABTS, NO scavenging, metal chelating, reducing power, total phenol, ascorbic acid, total antioxidant capacity) |
Leaf and bark extracts (acetone, ethanol, methanol, aqueous) |
Methanol leaf & bark: best DPPH, NO scavenging, reducing power; Ethanol bark: highest phenol & ascorbic acid; Ethanol leaf: highest total antioxidant capacity; metal chelating weak; overall strong antioxidant potential |
[74] |
The characteristic trait of diabetes mellitus, a group of complex metabolic disorders, is persistent hyperglycemia brought upon by either insulin resistance, a reduced production of insulin or both [75]. Ultimately, chronic hyperglycemia causes fatal problems in the liver, kidney and cardiovascular systems by inducing oxidative stress, glycation end product formation and carbohydrate metabolism dysregulation. Seeing the limitations and side effects that current synthetic antidiabetic agents can give, plant derived natural products are now in the highlights and are being increasingly investigated as an alternative or complementary therapy due to their quality to act on multiple biochemical pathways simultaneously [76].
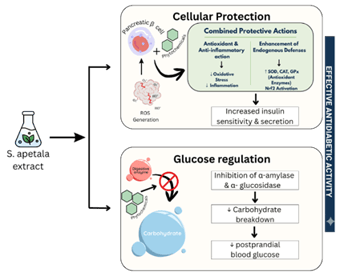
Figure 3: General antidiabetic mechanism of Sonneratia apetala extract
Sonneratia apetala has exhibited significant antidiabetic potential in both in vivo and in vitro models. In vitro models revealed that S. apetala extracts possessed potent inhibitory effects against carbohydrate digesting enzymes such as α-amylase and α-glucosidase, which delay the breakdown of carbohydrate and absorption of glucose [72, 74]. Furthermore, glucose uptake studies conducted using yeast models showed a dose dependent increase in the utilization of glucose, whereas glucose adsorption assays indicated that leaf extracts could bind to glucose molecules, consequently reducing their availability for absorption [77]. These findings together highlighted multiple mechanisms by which Sonneratia apetala can maintain or modulate blood glucose levels. In vivo studies further validated these outcomes as oral administration of the methanolic extract of fruit pericarp in streptozocin-induced diabetic rats, significantly reduced the fasting serum glucose over a prolonged treatment period [8]. Together these results suggest that both fruit and leaf derived extracts of S. apetala show hypoglycemic effects.
The antidiabetic activities of S. apetala observed across various experimental studies are summarized in Table 4, which shows the extract types, models used, and assays conducted as well as the key findings. Overall, the current evidence indicates that S. apetala holds a strong promise as a natural antidiabetic agent.
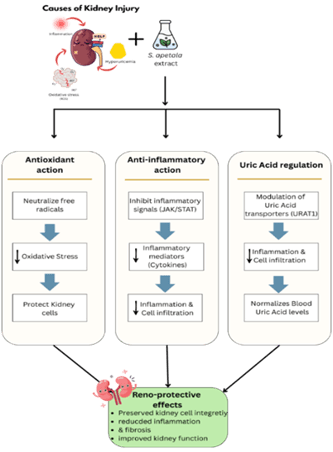
Kidney dysfunction and chronic kidney disease are often linked to underlying metabolic disorders such as diabetic nephropathy and conditions characterized by systemic oxidative stress and chronic inflammation [79]. Given that Keora possesses potent antioxidant and anti-inflammatory properties and significant anti-diabetic effects, it can be strongly positioned to have potential reno-protective agent. Even though research dedicated to its direct reno-protective effects is currently limited, the existing evidence is quite compelling [58, 68, 80].
A key study investigating the effects of the aqueous extract of S. apetala in a hyperuricemia mouse model showed robust evidence for its reno-protective potential. Hyperuricemia is one of the known causes of oxidative stress and inflammation in the kidney, often leading to kidney stone formation and renal injury. Treatment with the leaf extract showed restored antioxidant defenses (enhanced activity of antioxidant enzymes like SOD, CAT, GSH) and reduced oxidative stress markers like MDA and reactive oxygen species [58]. Similarly, seed oil extracts, branch and leaf extracts have also shown improvement in antioxidant activity, renal histology and uric acid transporters, further confirming the reno-protective potential of S. apetala [65, 77].
The general mechanism of reno-protective action is illustrated in Figure 4, and the specific experimental findings are summarized in Table 5.
Table 4 : Antidiabetic Effects of Sonneratia apetala Extracts
|
Model |
Treatment |
Result |
Ref. |
|
In-vivo. Male Long-EVANS- STZ-induced type 2 DM rats. |
Pericarp methanolic extract at 1.25g/10ml water/kg for 3 months. |
↓Reduced serum glucose level |
[8] |
|
In-vitro. α-amylase and α-glucosidase inhibition. |
10–1000 µg/mL (S. apetala fruit extract, in vitro) |
Showed strong inhibitory activity of α-amylase and α-glucosidase. |
[14] |
|
In vivo. Oral Glucose Tolerance Test (OGTT) in Swiss Albino Mice |
Methanolic extracts of 30 and 60 mg were administered; Blood samples were collected 120- and 180-min post-glucose administration. |
↓ Blood glucose levels; Significant antidiabetic potential. |
[64] |
|
In vitro. Yeast cells (Saccharomyces cerevisiae) |
Methanolic leaf extract of Sonneratia apetala (25–200 µg/mL) |
↑ Increased glucose uptake in a dose-dependent manner; highest uptake at 200 µg/mL |
[77] |
|
In vitro. α-Amylase enzyme assay |
Methanolic leaf extract of Sonneratia apetala (0.5–5 mg/mL) |
↓ Inhibited α-amylase; near 100% inhibition at 2 mg/mL |
[77] |
|
In vitro. Glucose adsorption assay |
Methanolic leaf extract of Sonneratia apetala (1%) |
↑ Adsorbed glucose proportionally to concentration; maximum at 100 mmol/L |
[77] |
|
In vivo. Mice (oral glucose tolerance test) |
Ethanolic extract of pneumatophores of Sonneratia apetala (250 & 500 mg/kg) |
↓ Blood glucose significantly at 60 and 120 min; |
[70] |
|
In vitro. α-Glucosidase inhibition |
Chloroform: Methanol (1:1) extract of leaves |
↓ Inhibited α-glucosidase activity (IC50 = 286 µg/mL) |
[78] |
|
In-vitro. α-amylase inhibition & Raphanus sativus root-growth inhibition |
Pneumatophore crude methanol extract, ethanol fraction (95%), acetone fraction (50% acetone) |
Crude: moderate α-amylase & root inhibition; Ethanol: low; Acetone: strong α-amylase & root inhibition (tannin-rich). |
[72] |
|
In vitro, Yeast α-glucosidase assay |
Leaf & bark extracts: Acetone, Ethanol, Methanol, Aqueous |
↓ Inhibited α-glucosidase activity in a dose-dependent manner; methanol extract most potent |
[74] |
Figure 4: Flowchart illustrating the general reno-protective mechanism of S. apetala extracts
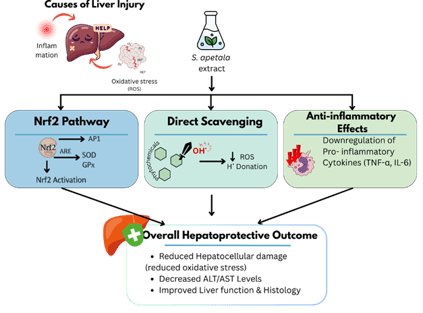
Figure 5: General Hepatoprotective mechanism of S. apetala Extract
Table 5 : Reno-protective effects of Sonneratia apetala extracts
|
Model |
|
Treatment |
|
Result |
|
Ref. |
|
In vivo. PO/HX-induced hyperuricemic mice. |
|
Aqueous extract of leaves, further concentrated with 60% ethanol |
|
Restored renal SOD, CAT, GSH-Px and reduced MDA and ROS in kidney tissue; ↓UA, BUN, CRE, Cys-C; |
|
[58] |
|
In vivo. Potassium oxonate/hypoxanthine-induced hyperuricemic mice |
|
Sonneratia apetala seed oil (SSO) |
|
↓ Serum UA, CRE, and BUN. ↑ SOD, CAT, GSH-Px with ↓ ROS and MDA levels. ↓ Kidney histopathological lesions. ↓ MCP-1, IL-1B, IL-6, IL-18, TNF-a. |
|
[80] |
|
In vivo. Hyperuricemia mice induced by Potassium oxonate (PO) and hypoxanthine (HX) |
|
Aqueous extract of leaves and branches |
|
↓ Kidney weight and index. ↓ Serum UA, CRE, and BUN. ↓ Kidney histopathological changes. ↓ MDA and ↑ CAT, SOD, and GSH. ↓ Renal inflammatory markers (IL-6, IL-18, IL-1ẞ, TNF-α, MCP-1, TGF-β). Regulated renal uric acid transporters (OAT1, URAT1, GLUT9). |
|
[68] |
|
In vivo. Isoprenaline-induced male Long-Evans rats |
|
Keora fruit peel extract 100 mg/kg/day |
|
↓MDA, NO, and AOPP levels in the kidney ↑CAT, SOD, and GSH activity in the kidney ↓MPO activity in the kidney ↓Creatinine and uric acid levels |
|
[81] |
Table 6: Hepatoprotective effects of Sonneratia apetala extracts
|
Model |
|
Treatment |
|
Result |
|
Ref. |
|
In-vivo. Male Kunming mice- acetaminophen-induced liver injury. |
|
Aqueous fruit extract at 100, 200, and 400mg/kg/day orally for 1 week. |
|
↑ Survival, ameliorated liver histology, ↓ ALT, AST, MDA, TNF-α, IL-6, MPO; ↑ GSH, GSH-Px, CAT, total antioxidant capacity |
|
[60] |
|
In vivo: Male Swiss albino mice with iron overload induced by ferric carboxymaltose |
|
In vivo: S. apetala fruit extract fractions (Hex, Chl, Met) at 100, 500, and 1,000 µg/kg bw daily for 15 days |
|
All fractions (Hex, Chl, Met) showed dose-dependent amelioration of iron overload. Met was most effective, completely ameliorating iron overload |
|
[61] |
|
In vivo (Kunming mice, HUA model) |
|
Aqueous extract of leaves and branches (50, 100, 200 mg/kg) for 7 days |
|
↓ UA, CRE, and BUN in serum. |
|
[68] |
|
In vivo. PO/HX-induced hyperuricemic mice. |
|
Aqueous extract of leaves, further concentrated with 60% ethanol |
|
No hepatotoxicity; ↓ hepatic XOD and ADA activities |
|
[58] |
Liver playing a crucial role in the metabolism and detoxification of substances like alcohol, drugs and metabolic byproducts makes it highly susceptible to injury and diseases, leading to conditions like NAFLD, fibrosis, or other metabolic disorders [82]. Hepatotoxicity is commonly associated with an elevated level of liver enzymes such as alanine aminotransferase (ALT) and aspartate aminotransferase (AST), lipid peroxidation, and inflammatory responses. Although synthetic hepatoprotective medications are available, prolonged use of such medicines may result in adverse side effects, which has led our researchers to the exploration of plant-derived natural products as alternatives or supplementary treatments [60, 61].
Sonneratia apetala has demonstrated significant hepatoprotective potential in various experimental models. In vivo studies using male Kunming mice with acetaminophen induced liver injury showed that the oral administration of the aqueous extract of fruit at 100, 200 and 400 mg/kg/day for 1 week significantly increased survival rats, ameliorated histological liver damage and also reduced the elevated levels of ALT, AST, malondialdehyde (MDA), TNF-α, IL-6 and myeloperoxidase (MPO). Alternatively, antioxidant markers including Glutathione (GSH), glutathione peroxidase (GSH-Px), catalase (CAT) and total antioxidant capacity were significantly increased, indicating a strong antioxidant and anti-inflammatory effects in the liver [60]. In addition to modulating oxidative stress, S. apetala extracts have also shown influence in purine metabolism by inhibiting hepatic xanthine oxidase (XOD) and adenosine deaminase (ADA), ultimately reducing the uric acid production and improving the renal-hepatic function in hyperuricemic mice models [58, 68]. Collectively, these findings portray the various ways S. apetala extracts can exert their protective effects on the liver. Figure 5 provides a comprehensive overview of S. apetala’s hepatoprotective effects. A comprehensive overview of different in vivo studies illustrating the hepatoprotective potential of S. apetala is provided in Table 6.
7. Toxicity and Safety
There is limited information regarding the toxicity and safety profile of Sonneratia apetala. Most of the in vivo study outcomes did not report any adverse outcomes, and the biochemical markers of hepatic and renal functions remained within normal ranges, suggesting tolerability at the tested doses [58, 60, 68]. However, a brine shrimp lethality bioassay showed cytotoxic potential in certain solvent fractions of the bark extract, highlighting that the extract may show toxicity based on the extraction method and plant part used [12]. Overall, while available data suggested relative safety in animal models, systemic acute, chronic, and subchronic toxicity studies are lacking. Future work should be done to address these gaps to establish a clear safety profile for potential therapeutic uses.
8. Future Perspective
This review provides a summary of the current scientific literature concerning the therapeutic potential of Sonneratia apetala (Keora). The findings showed that the diverse and potent pharmacological activities of this mangrove species are linked to its unique phytochemical composition [6]. The necessity for the plant to thrive under severe environmental stress has also driven it to evolve a robust defensive mechanism rich in polyphenols, flavonoids and triterpenoids [4]. The major pharmacological activities like antioxidant, antidiabetic, renoprotective and hepatoprotective effects for a unified therapeutic profile [3, 58]. The high antioxidant potential of Keora is its foundational activity acting as the molecular connection supporting its unique advantages against chronic diseases. For example, its robust antidiabetic and emerging reno-protective effects are not isolated activities but rather a direct consequence of its ability to mitigate oxidative stress and suppress chronic inflammation in tissues such as the liver and kidney [79]. The multi-targeting action of Keora, combining its anti-hyperglycemic effects with renal uric acid regulation, positions it as a promising candidate for managing metabolic syndrome and complications like diabetic nephropathy[58]. Despite these compelling preclinical findings, a major drawback is the limited research depth for certain effects. Therefore, future efforts must prioritize isolating lead compounds and validating these observations through further studies to fully understand their efficacy and safety profiles.
Although pharmacological evidence for Sonneratia apetala is promising, quite a large gap remains before it can be advanced towards clinical use. Most studies to date are limited to in vitro assays and small animal models, with little attention to pharmacokinetics, bioavailability, or long-term safety.
Future studies should thus focus on well-designed toxicity studies, analyses using modern molecular approaches and translational studies including clinical trials. Considering its rich and potent antioxidant and anti-inflammatory potential, evaluation in cardiovascular disease models such as myocardial infarction or cardiotoxicity models would also be valuable. With its broad phytochemical profile and efficacy against oxidative stress-driven disorders, S. apetala holds promise as a candidate for the development in Phyto-pharmaceutics.
9. Conclusion
Author Contributions
Funding
Data Availability Statement
Data used in this study will be available upon reasonable request from the corresponding author.
Acknowledgments
Conflicts of Interest
References
- Davis, C.C.; Choisy, P. Medicinal Plants Meet Modern Biodiversity Science. Curr. Biol. 2024, 34, R158–R173. https://doi.org/10.1016/j.cub.2023.12.038
- Chaachouay, N.; Douira, A.; Zidane, L. COVID-19, Prevention and Treatment with Herbal Medicine in the Herbal Markets of Salé Prefecture, North-Western Morocco. Eur. J. Integr. Med. 2021, 42, 101285. https://doi.org/10.1016/j.eujim.2021.101285
- Vaou, N.; Stavropoulou, E.; Voidarou, C.; Tsakris, Z.; Rozos, G.; Tsigalou, C.; Bezirtzoglou, E. Interactions between Medicinal Plant-Derived Bioactive Compounds: Focus on Antimicrobial Combination Effects. Antibiotics 2022, 11, 1014. https://doi.org/10.3390/antibiotics11081014
- Dahibhate, N.; Saddhe, A.; Kumar, K. Mangrove Plants as a Source of Bioactive Compounds: A Review. Nat. Prod. J. 2018, 8. https://doi.org/10.2174/2210315508666180910125328
- Nasrin, S.; Hossain, M.; Alam, M. A Monograph on Sonneratia apetala Buch.-Ham. 2017.
- Hossain, S.J.; Iftekharuzzaman, M.; Haque, M.A.; Saha, B.; Moniruzzaman, M.; Rahman, M.M.; Hossain, H. Nutrient Compositions, Antioxidant Activity, and Common Phenolics of Sonneratia apetala (Buch.-Ham.) Fruit. Int. J. Food Prop. 2016, 19, 1080–1092. https://doi.org/10.1080/10942912.2015.1055361
- Uddin, M.R.; Akhter, F.; Abedin, M.J.; Shaikh, M.A.A.; Al Mansur, M.A.; Rahman, M.S.; Molla Jamal, A.S.I.; Akbor, M.A.; Hossain, M.H.; Sharmin, S.; Idris, A.M.; Khandaker, M.U. Comprehensive Analysis of Phytochemical Profiling, Cytotoxic and Antioxidant Potentials, and Identification of Bioactive Constituents in Methanoic Extracts of Sonneratia apetala Fruit. Heliyon 2024, 10, e33507. https://doi.org/10.1016/j.heliyon.2024.e33507
- Hossain, S.J.; Basar, M.H.; Rokeya, B.; Arif, K.M.T.; Sultana, M.S.; Rahman, M.H. Evaluation of Antioxidant, Antidiabetic and Antibacterial Activities of the Fruit of Sonneratia apetala (Buch.-Ham.). Orient. Pharm. Exp. Med. 2013, 13, 95–102.
- Uddin, M.R.; Khandaker, M.U.; Akter, N.; Ahmed, M.F.; Hossain, S.M.M.; Gafur, A.; Abedin, M.J.; Rahman, M.A.; Idris, A.M. Identification and Economic Potentiality of Mineral Sands Resources of Hatiya Island, Bangladesh. Minerals 2022, 12, 1436. https://doi.org/10.3390/min12111436
- Bose, S.; Mazumder, S.; Ghosh, S.; Banerjee, S.; Roy, S.; Singh, N. Sonneratia apetala: Its Ecology, Bioactive Compounds and Biological Activities Including Its Nano-Formulations. J. Nat. Remedies 2023, 1287–1306. https://doi.org/10.18311/jnr/2023/34073
- Yi, X.; Jiang, S.; Qin, M.; Liu, K.; Cao, P.; Chen, S.; Deng, J.; Gao, C. Compounds from the Fruits of Mangrove Sonneratia apetala: Isolation, Molecular Docking and Antiaging Effects Using a Caenorhabditis elegans Model. Bioorg. Chem. 2020, 99, 103813. https://doi.org/10.1016/j.bioorg.2020.103813
- Hossain, S.J.; Islam, M.R.; Pervin, T.; Iftekharuzzaman, M.; Hamdi, O.A.A.; Mubassara, S.; Saifuzzaman, M.; Shilpi, J.A. Antibacterial, Anti-Diarrhoeal, Analgesic, Cytotoxic Activities, and GC–MS Profiling of Sonneratia apetala (Buch.-Ham.) Seed. Prev. Nutr. Food Sci. 2017, 22, 157–165. https://doi.org/10.3746/pnf.2017.22.3.157
- Ali, M. Keora or Mangrove Apple, Sonneratia apetala. Flora of Bangladesh 2025. Available online: https://www.floraofbangladesh.com/2019/08/keora-or-mangrove-apple.html
- Saha, A.; Id, T.; Seal, T.; Id, S. Characteristic Constituents with Antidiabetic Significance from Indian Mangrove Apples. Future Nat. Prod. 2023, 9, 3–9.
- Espíndola, K.M.M.; Ferreira, R.G.; Narvaez, L.E.M.; Rosario, A.C.R.S.; Silva, A.H.M.; Silva, A.G.B.; Vieira, A.P.O.; Monteiro, M.C. Chemical and Pharmacological Aspects of Caffeic Acid and Its Activity in Hepatocarcinoma. Front. Oncol. 2019, 9, 541. https://doi.org/10.3389/fonc.2019.00541
- Cikman, O.; Soylemez, O.; Ozkan, O.F.; Kiraz, H.A.; Sayar, I.; Ademoglu, S.; Taysi, S.; Karaayvaz, M. Antioxidant Activity of Syringic Acid Prevents Oxidative Stress in L-Arginine-Induced Acute Pancreatitis: An Experimental Study on Rats. Int. Surg. 2015, 100, 891–896. https://doi.org/10.9738/intsurg-d-14-00170.1
- Li, D.; Rui, Y.-X.; Guo, S.-D.; Luan, F.; Liu, R.; Zeng, N. Ferulic Acid: A Review of Its Pharmacology, Pharmacokinetics and Derivatives. Life Sci. 2021, 284, 119921. https://doi.org/10.1016/j.lfs.2021.119921
- Aldaba-Muruato, L.; Ventura-Juárez, J.; Perez-Hernandez, A.; Hernández-Morales, A.; Muñoz-Ortega, M.; Martínez-Hernández, S.; Alvarado-Sánchez, B.; Macías-Pérez, J. Therapeutic Perspectives of p-Coumaric Acid: Anti-Necrotic, Anti-Cholestatic and Anti-Amoebic Activities. World Acad. Sci. J. 2021, 3, 47. https://doi.org/10.3892/wasj.2021.118
- Kahkeshani, N.; Farzaei, F.; Fotouhi, M.; Alavi, S.S.; Bahramsoltani, R.; Naseri, R.; Momtaz, S.; Abbasabadi, Z.; Rahimi, R.; Farzaei, M.H.; Bishayee, A. Pharmacological Effects of Gallic Acid in Health and Diseases: A Mechanistic Review. Iran. J. Basic Med. Sci. 2019, 22, 225–237. https://doi.org/10.22038/ijbms.2019.32806.7897
- Prša, P.; Karademir, B.; Biçim, G.; Mahmoud, H.; Dahan, I.; Yalçın, A.S.; Mahajna, J.; Milisav, I. The Potential Use of Natural Products to Negate Hepatic, Renal and Neuronal Toxicity Induced by Cancer Therapeutics. Biochem. Pharmacol. 2020, 173, 113551. https://doi.org/10.1016/j.bcp.2019.06.007
- Emami, S.; Dadashpour, S. Current Developments of Coumarin-Based Anti-Cancer Agents in Medicinal Chemistry. Eur. J. Med. Chem. 2015, 102, 611–630. https://doi.org/10.1016/j.ejmech.2015.08.033
- Li, C.; Jing, Y.; Cheng, L.; Si, Z.; Mou, Z.; Niu, D.; Ma, F.; Liu, C. The Antifungal Activity of trans-Cinnamic Acid and Its Priming Effect in Apple in Response to Valsa mali. Plant Pathol. 2023, 72, 1595–1603. https://doi.org/10.1111/ppa.13786
- Shakil, S.; Zeeshan, M.; Khan, M.S.; Shaikh, S.; Biswas, D.; Ahmad, A.; Kamal, M.A.; Rizvi, S.M.D. An Enzoinformatics Study Targeting Polo-Like Kinase-1 Enzyme: Comparative Assessment of Anticancer Potential of Compounds Isolated from Leaves of Ageratum houstonianum. Pharmacogn. Mag. 2014, 10, S14–S21. https://doi.org/10.4103/0973-1296.127333
- Zou, H.; Ye, H.; Kamaraj, R.; Zhang, T.; Zhang, J.; Pavek, P. A Review on Pharmacological Activities and Synergistic Effect of Quercetin with Small Molecule Agents. Phytomedicine 2021, 92, 153736. https://doi.org/10.1016/j.phymed.2021.153736
- Jasemi, S.V.; Khazaei, H.; Morovati, M.R.; Joshi, T.; Aneva, I.Y.; Farzaei, M.H.; Echeverría, J. Phytochemicals as Treatment for Allergic Asthma: Therapeutic Effects and Mechanisms of Action. Phytomedicine 2024, 122, 155149. https://doi.org/10.1016/j.phymed.2023.155149
- Dehelean, C.A.; Coricovac, D.; Pinzaru, I.; Marcovici, I.; Macasoi, I.G.; Semenescu, A.; Lazar, G.; Pinzaru, S.C.; Radulov, I.; Alexa, E.; Cretu, O. Rutin Bioconjugates as Potential Nutraceutical Prodrugs: An In Vitro and In Ovo Toxicological Screening. Front. Pharmacol. 2022, 13, 1000608. https://doi.org/10.3389/fphar.2022.1000608
- Semwal, D.K.; Semwal, R.B.; Combrinck, S.; Viljoen, A. Myricetin: A Dietary Molecule with Diverse Biological Activities. Nutrients 2016, 8, 90. https://doi.org/10.3390/nu8020090
- Tao, T.; Zhang, P.; Zeng, Z.; Wang, M. Advances in Autophagy Modulation of Natural Products in Cervical Cancer. J. Ethnopharmacol. 2023, 314, 116575. https://doi.org/10.1016/j.jep.2023.116575
- Xue, J.-C.; Yuan, S.; Meng, H.; Hou, X.-T.; Li, J.; Zhang, H.-M.; Chen, L.-L.; Zhang, C.-H.; Zhang, Q.-G. The Role and Mechanism of Flavonoid Herbal Natural Products in Ulcerative Colitis. Biomed. Pharmacother. 2023, 158, 114086. https://doi.org/10.1016/j.biopha.2022.114086
- Uwemedimo, F., et al. Article no.AJBGMB.125799 Original Research Article Umoh et al. Asian Journal of Biochemistry Genetics and Molecular Biology. 2024;16:30-43.
- Alam, W.; Ahmed, I.; Ali, M.; Khan, F.; Khan, H. Chapter 8 – Neuroprotective Effect of Terpenoids. In Phytonutrients and Neurological Disorders; Khan, H., Aschner, M., Mirzaei, H., Eds.; Academic Press: London, UK, 2023; pp. 227–244.
- Youssef, A.M.M.; Maaty, D.A.M.; Al-Saraireh, Y.M. Phytochemical Analysis and Profiling of Antioxidants and Anticancer Compounds from Tephrosia purpurea (L.) subsp. apollinea (Fabaceae). Molecules 2023, 28. https://doi.org/10.3390/molecules28093939
- Amudha, P.; Jayalakshmi, M.; Pushpabharathi, N.; Vanitha, V. Identification of Bioactive Components in Enhalus acoroides Seagrass Extract by Gas Chromatography–Mass Spectrometry. Asian J. Pharm. Clin. Res. 2018, 11, 313–315.
- Demirci, B.; Demir, O.; Dost, T.; Birincioglu, M. Protective Effect of Vitamin B5 (Dexpanthenol) on Cardiovascular Damage Induced by Streptozocin in Rats. Bratisl. Lek. Listy 2014, 115, 190–196.
- Du, X.; Yang, Y.; Zhan, X.; Huang, Y.; Fu, Y.; Zhang, Z.; Liu, H.; Zhang, L.; Li, Y.; Wen, Q.; Zhou, X.; Zuo, D.; Zhou, C.; Li, L.; Hu, S.; Ma, L. Vitamin B6 Prevents Excessive Inflammation by Reducing Accumulation of Sphingosine-1-Phosphate in a Sphingosine-1-Phosphate Lyase-Dependent Manner. J. Cell Mol. Med. 2020, 24, 13129–13138. https://doi.org/10.1111/jcmm.15917
- Prakash, M.; Basavaraj, B.V.; Chidambara Murthy, K.N. Biological Functions of Epicatechin: Plant Cell to Human Cell Health. J. Funct. Foods 2019, 52, 14–24. https://doi.org/10.1016/j.jff.2018.10.021
- Lescano, C.H.; Freitas de Lima, F.; Caires, A.R.L.; de Oliveira, I.P. Chapter 25 – Polyphenols Present in Campomanesia Genus: Pharmacological and Nutraceutical Approach. In Polyphenols in Plants, 2nd ed.; Watson, R.R., Ed.; Academic Press: London, UK, 2019; pp. 407–420. https://doi.org/10.1016/B978-0-12-813768-0.00027-X
- Uddin, M.R.; Akhter, F.; Abedin, M.J.; Shaikh, M.A.A.; Al Mansur, M.A.; Rahman, M.S.; Molla Jamal, A.S.I.; Akbor, M.A.; Hossain, M.H.; Sharmin, S.; Idris, A.M.; Khandaker, M.U. Comprehensive Analysis of Phytochemical Profiling, Cytotoxic and Antioxidant Potentials, and Identification of Bioactive Constituents in Methanoic Extracts of Sonneratia apetala Fruit. Heliyon 2024, 10, e33507. https://doi.org/10.1016/j.heliyon.2024.e33507
- Holanda, L.E.G.d.; Ramos, C.d.S.; Freitas Filho, J.R.d. Fatty Acid Profiles and Antimicrobial Activity from Tropical Fruit Seeds. J. Mex. Chem. Soc. 2023, 67, 163–171. https://doi.org/10.29356/jmcs.v67i2.1866
- Ahmed, A.; Ohlson, M.; Hoque, S.; Moula, M.G. Chemical Composition of Leaves of a Mangrove Tree (Sonneratia apetala Buch.-Ham.) and Their Correlation with Some Soil Variables. Bangladesh J. Bot. 2010, 39, 61–69. https://doi.org/10.3329/bjb.v39i1.5528
- Shoge, M.; Amusan, T. Phytochemical, Antidiarrhoeal Activity, Isolation and Characterisation of 11-Octadecenoic Acid, Methyl Ester Isolated from the Seeds of Acacia nilotica Linn. J. Biotechnol. Immunol. 2020, 2, 1–12.
- Pinto, M.E.A.; Araújo, S.G.; Morais, M.I.; Sá, N.P.; Lima, C.M.; Rosa, C.A.; Siqueira, E.P.; Johann, S.; Lima, L.A.R.S. Antifungal and Antioxidant Activity of Fatty Acid Methyl Esters from Vegetable Oils. An. Acad. Bras. Cienc. 2017, 89, 1671–1681. https://doi.org/10.1590/0001-3765201720160908
- Fagali, N.; Catalá, A. Antioxidant Activity of Conjugated Linoleic Acid Isomers, Linoleic Acid and Its Methyl Ester Determined by Photoemission and DPPH Techniques. Biophys. Chem. 2008, 137, 56–62. https://doi.org/10.1016/j.bpc.2008.07.001
- Du, J.; Zhao, L.; Ma, L.-Q.; Liu, Y.-B.; Shi, G.-L.; Wang, Y.-N.; Tong, B.-S. Acaricidal Activity of the Oleic Acid Methyl Ester from Pharbitis purpurea Seeds against Tetranychus cinnabarinus. In Information Technology and Agricultural Engineering; Zhu, E., Sambath, S., Eds.; Springer: Berlin, Heidelberg, 2012; pp. 621–628.
- Alabi, K.; Lajide, L.; O., J. Biological Activities of Oleic Acid and Its Primary Amide: Experimental and Computational Studies. J. Chem. Soc. Niger. 2018, 43, 38–49.
- Tsoupras, A.; Cholidis, P.; Kranas, D.; Galouni, E.A.; Ofrydopoulou, A.; Efthymiopoulos, P.; Shiels, K.; Saha, S.K.; Kyzas, G.Z.; Anastasiadou, C. Anti-Inflammatory, Antithrombotic, and Antioxidant Properties of Amphiphilic Lipid Bioactives from Shrimp. Pharmaceuticals 2024, 18, 25. https://doi.org/10.3390/ph18010025
- Cholidis, P.; Kranas, D.; Chira, A.; Galouni, E.A.; Adamantidi, T.; Anastasiadou, C.; Tsoupras, A. Shrimp Lipid Bioactives with Anti-Inflammatory, Antithrombotic, and Antioxidant Health-Promoting Properties for Cardio-Protection. Mar. Drugs 2024, 22, 554. https://doi.org/10.3390/md22120554
- Wang, Z.-J.; Li, G.-M.; Nie, B.-M.; Lu, Y.; Yin, M. Neuroprotective Effect of Stearic Acid against Oxidative Stress via Phosphatidylinositol 3-Kinase Pathway. Chem. Biol. Interact. 2006, 160, 80–87. https://doi.org/10.1016/j.cbi.2005.12.008
- Altinoz, M.A.; Ozpinar, A. PPAR-δ and Erucic Acid in Multiple Sclerosis and Alzheimer’s Disease: Likely Benefits in Terms of Immunity and Metabolism. Int. Immunopharmacol. 2019, 69, 245–256. https://doi.org/10.1016/j.intimp.2019.01.057
- Save, S. Determination of 1,2-Benzenedicarboxylic Acid, Bis(2-ethylhexyl) Ester from the Twigs of Thevetia peruviana as a Colwell Biomarker. J. Innov. Pharm. Biol. Sci. 2015, 2, 349–362.
- Sujatha, S.; Anand, S.; Sangeetha, K.N.; Shilpa, K.; Lakshmi, J.; Balakrishnan, A.; Lakshmi, B.S. Biological Evaluation of (3β)-Stigmast-5-en-3-ol as a Potent Anti-Diabetic Agent in Regulating Glucose Transport Using an In Vitro Model. Int. J. Diabetes Mellit. 2010, 2, 101–109. https://doi.org/10.1016/j.ijdm.2009.12.013
- Lou, H.; Li, H.; Zhang, S.; Lu, H.; Chen, Q. A Review on Preparation of Betulinic Acid and Its Biological Activities. Molecules 2021, 26. https://doi.org/10.3390/molecules26185583
- Xiong, Y.; Ren, L.; Wang, Z.; Hu, Z.; Zhou, Y. Anti-Proliferative Effect of Physcion on Human Gastric Cell Line via Inducing ROS-Dependent Apoptosis. Cell Biochem. Biophys. 2015, 73, 537–543. https://doi.org/10.1007/s12013-015-0674-9
- Davis, R.H.; Maro, N.P. Aloe vera and Gibberellin: Anti-Inflammatory Activity in Diabetes. J. Am. Podiatr. Med. Assoc. 1989, 79, 24–26. https://doi.org/10.7547/87507315-79-1-24
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxid. Med. Cell. Longev. 2017, 2017, 8416763. https://doi.org/10.1155/2017/8416763
- Mukherjee, S.; Chopra, H.; Goyal, R.; Jin, S.; Dong, Z.; Das, T.; Bhattacharya, T. Therapeutic Effect of Targeted Antioxidant Natural Products. Discover Nano 2024, 19, 144. https://doi.org/10.1186/s11671-024-04100-x
- Mukul, M.E.H.; Hossain, M.S.; Ahamed, S.K.; Debnath, P.; Akter, M. Antioxidant and Membrane Stabilizing Activities of Bark of Sonneratia apetala. Bangladesh Pharm. J. 2016, 19, 147–151. https://doi.org/10.3329/bpj.v19i2.29272
- Jiang, L.; Wu, Y.; Qu, C.; Lin, Y.; Yi, X.; Gao, C.; Cai, J.; Su, Z.; Zeng, H. Hypouricemic Effect of Gallic Acid, a Bioactive Compound from Sonneratia apetala Leaves and Branches, on Hyperuricemic Mice. Food Funct. 2022, 13, 10275–10290. https://doi.org/10.1039/d2fo02068h
- Leopoldini, M.; Russo, N.; Toscano, M. The Molecular Basis of the Working Mechanism of Natural Polyphenolic Antioxidants. Food Chem. 2011, 125, 288–306. https://doi.org/10.1016/j.foodchem.2010.08.012
- Liu, J.; Luo, D.; Wu, Y.; Gao, C.; Lin, G.; Chen, J.; Wu, X.; Zhang, Q.; Cai, J.; Su, Z. The Protective Effect of Sonneratia apetala Fruit Extract on Acetaminophen-Induced Liver Injury in Mice. Evid.-Based Complement. Altern. Med. 2019, 2019, 6919834.
- Mithila, M.; Islam, M.R.; Khatun, M.R.; Gazi, M.S.; Hossain, S.J. Sonneratia apetala (Buch.-Ham.) Fruit Extracts Ameliorate Iron Overload and Iron-Induced Oxidative Stress in Mice. Prev. Nutr. Food Sci. 2023, 28, 278–284. https://doi.org/10.3746/pnf.2023.28.3.278
- Bulbul, I.J.; Koly, S.F.; Begum, Y.; Jahan, N.; Nahar, N.; Islam, M.S. Assessment of Antimicrobial, Cytotoxic and Antioxidant Potentials of Sonneratia apetala Fruits. Biomed. Pharmacol. J. 2025, 18, 581–591. https://doi.org/10.13005/bpj/3110
- Nguyễn, T.B.T.; Dương, T.M.D.; Ngô, T.T.H.; Nguyễn, K.N.; Hoàng, T.P.L. Evaluation of Antioxidant and Gastroprotective Activities of Sonneratia apetala Leaf Extract. J. Sci. Technol. 2025, 8, 10–18. https://doi.org/10.55401/wgxs2x28
- Akhter, F.; Uddin, M.R.; Al Mansur, M.A.; Rahman, M.S.; Akbor, M.A.; Akter, N.; Idris, A.M.; Mostafa, M.G.; Molla Jamal, A.S.I.; Kamruzzaman, S. Antidiarrheal, Analgesic, Antidepressant, Antimicrobial and Hypoglycemic Activities of Sonneratia apetala Fruit Extract. PLoS ONE 2025, 20, e0321280.
- Murugesan, M.; Kandhavelu, M.; Thiyagarajan, R.; Natesan, S.; Rajendran, P.; Murugesan, A. Marine Halophyte-Derived Polyphenols Inhibit Glioma Cell Growth through MAPK Signaling Pathway. Biomed. Pharmacother. 2023, 159, 114288. https://doi.org/10.1016/j.biopha.2023.114288
- Khatun, M.R.; Luna, N.I.; Akter, S.; Islam, M.R.; Hossain, S.J. Antioxidant Activity and Capacity of Silver Nanoparticles Biosynthesized from Common Fruits of the Sundarban Forest. Khulna Univ. Stud. 2022, 19, 66–73. https://doi.org/10.53808/KUS.2022.19.01.2208-ls
- Sur, T.K.; Hazra, A.; Bhattacharyya, D.; Hazra, A.K. Antiradical and Antiulcer Effect of Sonneratia apetala Leaves against Alcohol-Induced Gastric Injury. Ars Pharm. 2021, 62, 348–357. https://doi.org/10.30827/ars.v62i3.20759
- Wu, Y.-L.; Chen, J.-F.; Jiang, L.-Y.; Wu, X.-L.; Liu, Y.-H.; Gao, C.-J.; Wu, Y.; Yi, X.-Q.; Su, Z.-R.; Cai, J.; Chen, J.-N. Sonneratia apetala Leaf and Branch Extract Ameliorates Hyperuricemia via JAK/STAT Pathway Suppression. Front. Pharmacol. 2021, 12, 698219. https://doi.org/10.3389/fphar.2021.698219
- Sarkar, K.; Das, A.; Modak.; Halder.; Islam, T.; Chowdhury.; Kundu, K.; Sarkar, B. Phytochemical Screening and Antioxidant Profiling of Two Mangrove Species of Sundarbans: Heritiera fomes and Sonneratia apetala. 2020.
- Biswas, B.; Golder, M.; Islam, T.; Sadhu, S.K. Comparative Antioxidative and Antihyperglycemic Profiles of Two Mangrove Species. Dhaka Univ. J. Pharm. Sci. 2018, 17, 205–211. https://doi.org/10.3329/dujps.v17i2.39177
- Nagababu, P.; Rao, V.U. Pharmacological Assessment and Green Synthesis of Silver Nanoparticles from Sonneratia apetala. J. Appl. Pharm. Sci. 2017, 7, 175–182. https://doi.org/10.7324/JAPS.2017.70824
- Mai, S.; Van Tan, D. Fractionation of Phenolic Compounds from Sonneratia apetala Pneumatophores and Their Bioactivities. Acad. J. Biol. 2017, 39, 451–456. https://doi.org/10.15625/0866-7160/v39n4.10708
- Hossain, S.; Pervin, T.; Suma, S. Effects of Cooking Methods on Phenolics and Antioxidant Activity of Sonneratia apetala Fruits. Int. Food Res. J. 2016, 23.
- Patra, J.K.; Das, S.K.; Thatoi, H. Phytochemical Profiling and Bioactivity of Mangrove Plant Sonneratia apetala. Chin. J. Integr. Med. 2015, 21, 274–285. https://doi.org/10.1007/s11655-014-1854-y
- Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 2009, 32, S62–S67. https://doi.org/10.2337/dc09-S062
- Rahman, M.M.; Dhar, P.S.; Anika, F.; Ahmed, L.; Islam, M.R.; Sultana, N.A.; Cavalu, S.; Pop, O.; Rauf, A. Exploring Plant-Derived Bioactive Substances as Antidiabetic Agents. Biomed. Pharmacother. 2022, 152, 113217. https://doi.org/10.1016/j.biopha.2022.113217
- Sarkar, B.; Barman, S.K.; Akhter, S.; Akter, R.; Das, J.; Sarkar, A.P.; Akter, R.; Kundu, S. Evaluation of In Vitro Anti-Diabetic Activity of Two Mangrove Plant Extracts: Heritiera fomes and Sonneratia apetala. J. Pharmacogn. Phytochem. 2019, 8, 2376–2380. https://doi.org/10.13140/RG.2.2.14398.72002
- Martinez-Luis, S.; Lopez, D.; Cherigo, L.; de Sedas, A.; Spadafora, C. Evaluation of Antiparasitic, Anticancer, Antimicrobial and Hypoglycemic Properties of Mangrove Plants. Asian Pac. J. Trop. Med. 2018, 11. https://doi.org/10.4103/1995-7645.223531
- Podkowińska, A.; Formanowicz, D. Chronic Kidney Disease as Oxidative Stress- and Inflammation-Mediated Cardiovascular Disease. Antioxidants 2020, 9. https://doi.org/10.3390/antiox9080752
- Chen, J.; Xu, L.; Jiang, L.; Wu, Y.; Wei, L.; Wu, X.; Xiao, S.; Liu, Y.; Gao, C.; Cai, J.; Su, Z. Sonneratia apetala Seed Oil Attenuates Hyperuricemia and Renal Injury in Mice. Food Funct. 2021, 12, 9416–9431. https://doi.org/10.1039/d1fo01830b
- Sultana, S.; Alimullah, M.; Ghosh, H. C.; Jalal, T.; Hossen, M. T.; Joya, A. B.; Akter, A.; Akhter, T.; Alauddin, J.; Subhan, N., Alam, M. A. Mangrove Fruit Keora Peel Extract Prevents Oxidative Stress and Fibrosis in the Kidney of ISO Administered Rats. J. Bio. Exp. Pharm. 2025, 3(1), 89-100.
- Chaudhary, N.; Arif, M.; Shafi, S.; Kushwaha, S.P.; Soni, P. Emerging Role of Natural Bioactive Compounds in Navigating the Future of Liver Disease. iLIVER 2025, 4, 100140. https://doi.org/10.1016/j.iliver.2024.100140
Figures
Author Affiliation
1Department of Pharmaceutical Sciences, North South University, Dhaka, Bangladesh
2School of Pharmacy, Brac University, Dhaka, Bangladesh
ARTICLE INFO
Dr. Raushanara Akter, Professor, School of Pharmacy, Brac University, Bangladesh
Dr. Md. Ashraful Alam, Professor, Department of Pharmaceutical Sciences, North South University, Bangladesh


-png.png?width=109&height=83&name=Phenol%2c%203%2c5-bis(1%2c1-dimethylethyl)-png.png)




%20ester-png.png?width=1789&height=455&name=bis(2-ethylhexyl)%20ester-png.png)